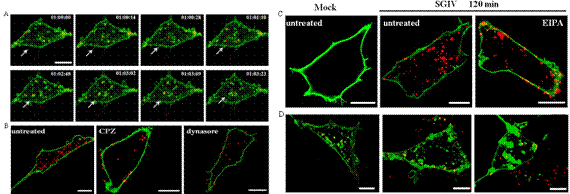
A research team led by Prof. Qin Qiwei from South China Sea Institute of Oceanology, collaborated with another research team led by Prof. Wang Hongda from Changchun Institute of Applied Chemistry, firstly applied single particle tracking technology in exploring the infection mechanism of fish DNA viruses. The research results have been published in Journal of virology (2014,88(22),13047-13063).
Viruses can only replicate in host cells and develop many strategies for successful infection. Investigation of the mechanism of virus entry will provide new insights into understanding of virology and cell biology. Iridoviruses are nucleocytoplasmic DNA viruses which cause great economic losses in the aquaculture industry but also show significant threat to global biodiversity. However, a lack of host cells has resulted in poor understanding of iridovirus behavior. Singapore grouper iridovirus (SGIV) is a novel marine fish DNA virus which belongs to genus Ranavirus, family Iridoviridae. The researchers investigated the crucial events during virus entry using a combination of single-virus tracking and biochemical assays, based on the established virus-cell infection model for SGIV. They observed that SGIVs could travel along filopodia-like protrusions and entered into the cells, by tracking individual SGIV particles in real time. Furthermore, making use of single particle imaging, immunofluorescence microscope and western blotting, they demonstrated that SGIV entered grouper cells via the clathrin-mediated endocytic pathway in a pH-dependent manner but not via caveola-dependent endocytosis, and proposed for the first time that macropinocytosis was involved in iridovirus entry. In addition, they monitored the transport of single virus particles in live cells, and found that SGIV depended on microtubules or actin filaments for intracellular transport. This work not only contributes greatly to understating iridovirus pathogenesis but also provides an ideal model for exploring the behavior of DNA viruses in living cells.
The work is supported by National Natural Science Foundation of China and the National Basic Research Program of China (973 Program).